Introduction
Antimicrobial resistance among Gram-negative bacteria has emerged as one of the most serious threats to clinical practice. Among these organisms, carbapenem-resistant Enterobacterales (CRE) are particularly important. CRE infections are associated with limited therapeutic options, prolonged hospitalization, increased healthcare costs and increased morbidity and mortality. [1] The 2024 World Health Organization (WHO) bacterial priority pathogens list classifies CRE among the critical-priority pathogens underscoring the urgent need for effective therapeutic strategies. Bloodstream infections caused by resistant Escherichia coli are challenging because they may occur in patients with critical illness, advanced age, or immunocompromise and may rapidly progress to sepsis and multiorgan dysfunction. [2]
A particularly problematic subgroup of CRE comprises organisms producing metallo-β-lactamases (MBLs), especially New Delhi metallo-β-lactamase (NDM). These enzymes hydrolyze nearly all β-lactam antibiotics, including carbapenems. They are not inhibited by currently available β-lactamase inhibitors such as Avibactam, vaborbactam, or Relebactam when used in their conventional combinations. Although Aztreonam is structurally stable against hydrolysis by metallo-β-lactamases, its activity is often compromised because NDM-producing organisms frequently co-produce other β-lactamases, including extended-spectrum β-lactamases and AmpC enzymes, that are capable of hydrolyzing Aztreonam. Consequently, infections caused by NDM-producing Enterobacterales remain among the most difficult resistant infections to treat and often require the use of older agents with important toxicity limitations.[3]
Combination therapy using Aztreonam with a β-lactamase inhibitor has emerged as a rational therapeutic strategy for metallo-β-lactamase-producing organisms. The best-known example is Ceftazidime-Avibactam plus Aztreonam, in which Avibactam inhibits co-produced serine β-lactamases thereby preserving Aztreonam activity. A similar mechanistic rationale applies to the combination of Imipenem-Cilastatin-Relebactam plus Aztreonam. In this regimen, Aztreonam retains intrinsic activity against NDM whereas Relebactam inhibits class A and class C β-lactamases that would otherwise hydrolyze Aztreonam. This pharmacologic synergy may restore Aztreonam activity in selected isolates co-producing metallo-β-lactamases and additional serine β-lactamases. In vitro studies have shown synergistic and bactericidal activity of Aztreonam combined with Imipenem-Relebactam against Enterobacterales co-producing NDM and additional serine β-lactamases.[4]
Despite this promising microbiological rationale, clinical evidence supporting Imipenem-Cilastatin-Relebactam plus Aztreonam remains limited. Most published guidance and clinical experience have focused on Ceftazidime-Avibactam plus Aztreonam whereas only a small number of reports have described the use of Relebactam-based regimens in this setting. This issue has become increasingly relevant as resistance to Ceftazidime-Avibactam-containing regimens, including Ceftazidime-Avibactam plus Aztreonam, is being recognized in selected isolates with complex resistance mechanisms. The 2024 Infectious Diseases Society of America (IDSA) guidance supports Aztreonam-containing combinations for infections caused by metallo-β-lactamase-producing Enterobacterales while acknowledging the limited clinical outcome data available for some regimens.[5]
There remains an important knowledge gap regarding the clinical effectiveness of Imipenem-Cilastatin-Relebactam plus Aztreonam for bloodstream infections caused by NDM-producing Escherichia coli. This is particularly relevant when isolates are resistant to Ceftazidime-Avibactam and Ceftazidime-Avibactam plus Aztreonam. Additional uncertainty exists regarding the performance of this regimen in markedly different host settings including profound immunosuppression, extremes of age, renal dysfunction and critical illness. In this report, we describe two cases of Ceftazidime-Avibactam–Aztreonam-resistant NDM-producing Escherichia coli bacteremia in which microbiological clearance was achieved after treatment with Imipenem-Cilastatin-Relebactam plus Aztreonam. These cases add to the emerging clinical literature supporting this combination as a potential salvage option for extensively drug-resistant Enterobacterales.
Case presentation
CASE 1
A 59-year-old man was admitted with a one week history of fever, fatigue and shortness of breath. He had a history of chronic hepatitis C infection, heavy tobacco use (exceeding 30 pack years) and previous lumbar spinal surgery. Three months before admission, routine laboratory evaluation had shown mild anemia with a hemoglobin concentration of 11 g/dL. There was no known family history of hematologic malignancies.The patient presented acutely unwell with fever and hemodynamic instability. Laboratory investigations showed markedly elevated inflammatory markers and blast cells on peripheral smear. Subsequent bone marrow biopsy confirmed AML. Molecular profiling confirmed a FLT3-ITD mutation and NPM1 co-mutation (Table 1).
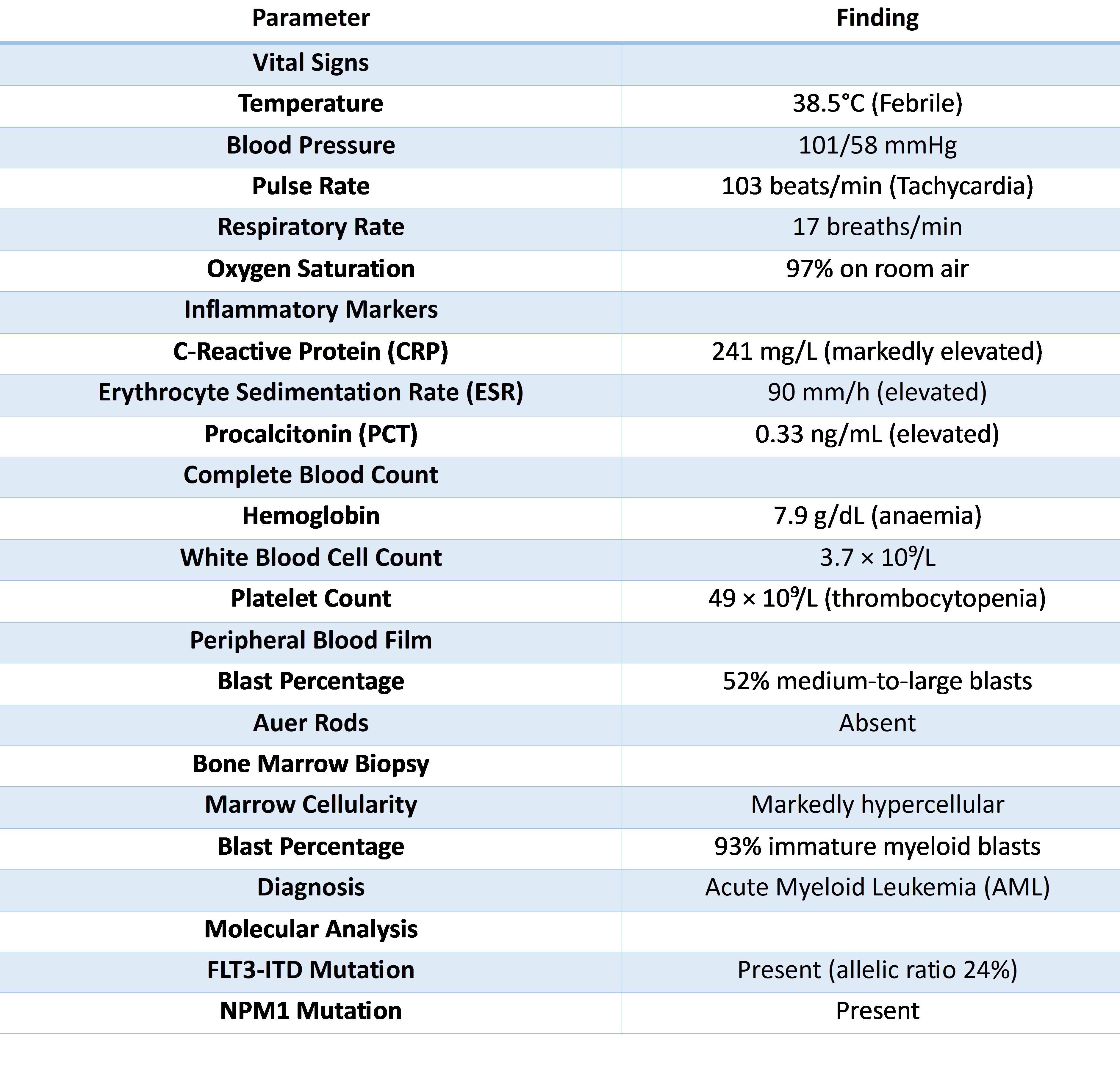
In view of suspected hematologic sepsis, empiric antimicrobial therapy with Piperacillin-Tazobactam and Levofloxacin was initiated. Blood cultures and sensitivity showed carbapenem-resistant Escherichia coli. Molecular resistance testing detected New Delhi metallo-β-lactamase. Antimicrobial susceptibility testing showed resistance to nearly all antimicrobial agents. This included resistance to Ceftazidime-Avibactam and the combination of Ceftazidime-Avibactam plus Aztreonam. The isolate was found to be susceptible only to Colistin, Gentamicin, and Tobramycin.Subsequently, treatment was escalated to Ceftazidime-Avibactam plus Aztreonam, together with Meropenem and Colistin. However, the patient remained febrile and inflammatory markers continued to rise, suggesting persistent uncontrolled infection. In view of persistent infection not responding to therapy, antimicrobial therapy was changed to Imipenem-Cilastatin-Relebactam plus Aztreonam, in combination with Colistin and Gentamicin. Subsequent blood cultures obtained on four further occasions remained negative, confirming microbiological clearance. Inflammatory markers gradually improved, although renal function worsened during therapy, most likely as a consequence of nephrotoxic antimicrobial exposure.Despite successful eradication of the bloodstream infection, the patient's overall condition deteriorated because of complications related to his underlying hematologic malignancy.
He subsequently succumbed during the same hospitalization. Importantly, repeat blood cultures remained negative for NDM-producing Escherichia coli. Patient's death was attributed to leukemia-related complications rather than persistent bacteremia.
CASE 2
A 91-year-old woman with a history of chronic obstructive pulmonary disease (COPD) requiring home bilevel positive airway pressure support, Alzheimer disease, diabetes mellitus, and chronic kidney disease (CKD) was electively admitted after a respiratory panel was reported positive for influenza A, Pseudomonas aeruginosa and Serratia marcescens. She presented with a 3-day history of fever and worsening cough. Inflammatory markers were found to be mildly elevated. Blood count revealed a mild anaemia with an otherwise preserved white cell and platelet count. The overall picture suggested an infective or inflammatory process without evidence of haematological malignancy on initial workup (Table 2).
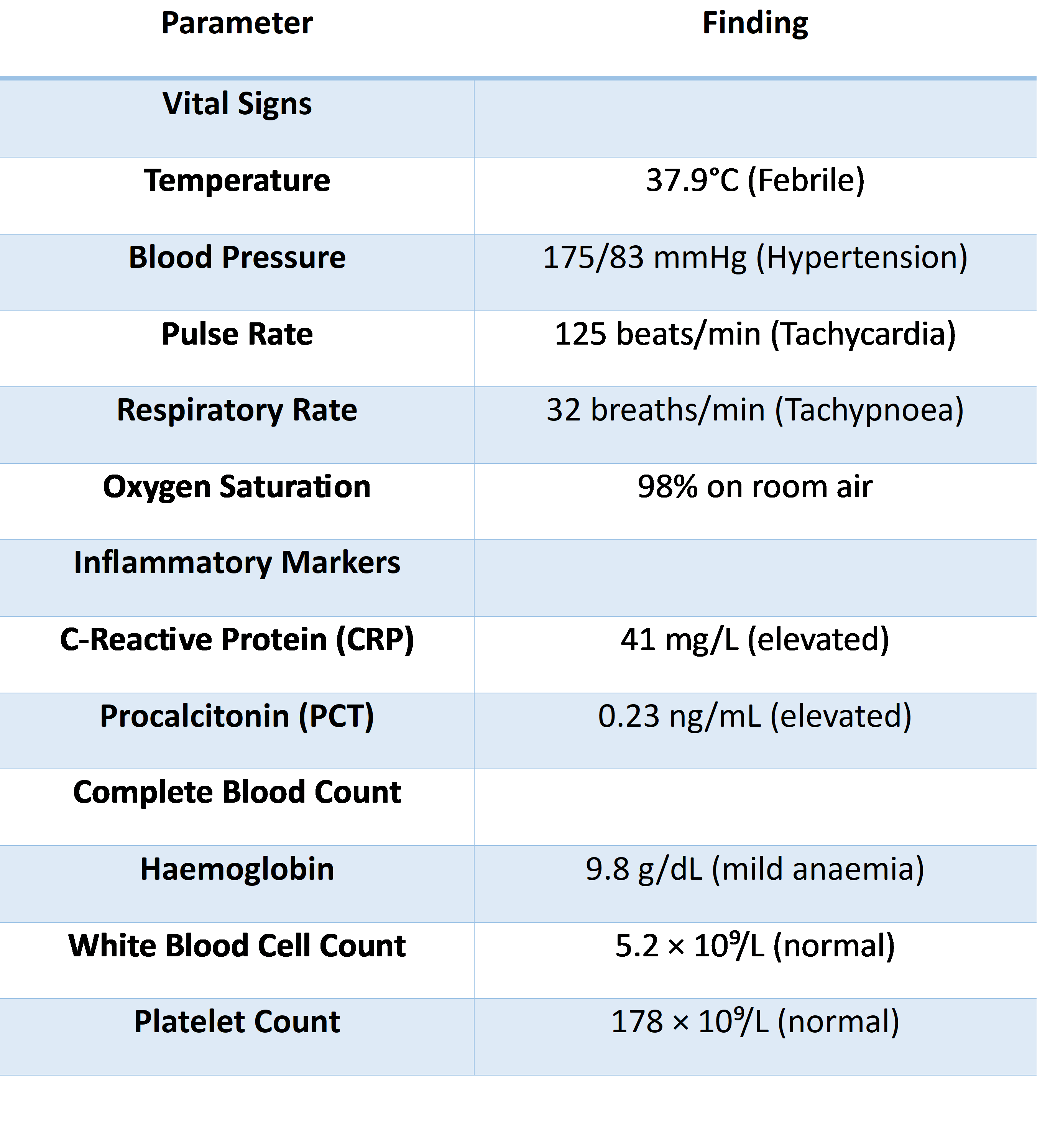
During her hospital stay she developed type 2 respiratory failure with reduced level of consciousness, requiring endotracheal intubation and transfer to the intensive care unit (ICU). Sputum culture subsequently grew a carbapenemase-producing Escherichia coli. A respiratory molecular panel confirmed the presence of Escherichia coli harboring both New Delhi metallo-β-lactamase and Cefotaximase-Munich (CTX-M) resistance genes. Later, a blood culture obtained also yielded the same NDM- and CTX-M-producing Escherichia coli. Antimicrobial susceptibility testing demonstrated resistance to nearly all tested antibiotics, including Ceftazidime-Avibactam and Ceftazidime-Avibactam plus Aztreonam, with retained susceptibility only to Colistin, Gentamicin, and Amikacin.Following the positive blood culture, antimicrobial therapy was changed to the combination of Imipenem-Cilastatin-Relebactam and Aztreonam. Because of underlying renal impairment, both agents were dose-adjusted and administered as Imipenem-Cilastatin-Relebactam 0.5 g intravenously every 6 hours and Aztreonam 1 g intravenously every 6 hours. This regimen was associated with a rapid clinical and microbiological response. Follow-up blood cultures obtained were negative. In parallel, procalcitonin, which had peaked at 3.48 ng/mL, decreased to 0.8 ng/mL within 3 days and continued to improve thereafter.The patient was eventually weaned from invasive mechanical ventilation, although she remained tracheostomy-dependent. After completion of a 7-day course of combination therapy she was discharged in stable condition and was transferred to a long-term care facility.
Discussion
This case series highlights the potential clinical utility of Imipenem-Cilastatin-Relebactam plus Aztreonam as a salvage regimen for bloodstream infection caused by New Delhi metallo-β-lactamase-producing Escherichia coli that was resistant not only to conventional agents but also to Ceftazidime-Avibactam and Ceftazidime-Avibactam plus Aztreonam. Microbiological clearance was achieved in both patients after initiation of this combination, despite markedly different host factors, including profound immun-ocompromise in one case and extreme frailty with chronic organ dysfunction in the other. These observations are clinically important because therapeutic options for metallo-β-lactamase-producing carbapenem-resistant Entero- bacterales remain extremely limited, particularly when front-line Aztreonam-based strategies are no longer active.[6]
The mechanistic rationale for this regimen is biologically compelling. Aztreonam remains stable against hydrolysis by metallo-β-lactamases, whereas Relebactam inhibits accompanying class A and class C serine β-lactamases that would otherwise hydrolyze Aztreonam. Although Relebactam does not directly inhibit New Delhi metallo-β-lactamase, its ability to protect Aztreonam in isolates co-producing extended-spectrum β-lactamases or AmpC enzymes provides the basis for the observed synergy. This concept is supported by the in vitro work of Biagi et al, who demonstrated that Aztreonam combined with Imipenem-Relebactam retained activity against clinical and isogenic strains of Escherichia coli and Klebsiella pneumoniae co-producing New Delhi metallo-β-lactamase and serine β-lactamases.[7]
Our findings should also be interpreted in the context of the limited but growing clinical literature. Pipitò et al described the first reported successful use of Imipenem-Relebactam plus Aztreonam in a patient with multidrug-resistant Klebsiella pneumoniae sternal infection complicated by bacteremia, thereby providing proof of concept for this regimen in severe invasive infection.[8] In contrast, Simner et al reported a case of New Delhi metallo-β-lactamase-producing Escherichia coli with resistance to all currently available β-lactam options, including Cefiderocol and Ceftazidime-Avibactam plus Aztreonam, underscoring the rapid evolution of near-pan-β-lactam resistance in this species.[9]
Likewise, Senchyna et al documented sequential treatment failure with Aztreonam plus Ceftazidime-Avibactam followed by Cefiderocol in bacteremic New Delhi metallo-β-lactamase-producing Escherichia coli, demonstrating how therapeutic pressure may select for highly resistant subpopulations during the course of infection.[10] Together, these reports by Pipitò, Simner, and Senchyna frame our cases within an emerging clinical reality in which treatment of New Delhi metallo-β-lactamase-producing Escherichia coli is increasingly threatened by adaptive and acquired resistance.An important strength of the present report is that both patients had documented bloodstream infection with microbiological clearance after introduction of the Imipenem-Cilastatin-Relebactam plus Aztreonam combination. In the first case, clearance occurred despite active acute myeloid leukemia and severe systemic illness, as well as prior clinical failure of a regimen containing Ceftazidime-Avibactam plus Aztreonam. In the second case, clearance was achieved in an elderly critically ill patient with chronic kidney disease and prolonged intensive care unit exposure. These contrasting host settings suggest that the regimen may have utility across a broad clinical spectrum.[11] In the first case in particular, concomitant use of Colistin and Gentamicin should be acknowledged as a potential confounding factor when attributing response to the combination regimen.
From a therapeutic standpoint, our experience is aligned with the broader principles of the Infectious Diseases Society of America guidance, which supports Aztreonam-containing combinationsformetallo-β-lactamase-producing Enterobacteraleswhileacknowledgingthathigh-quality comparative clinical data remain limited. However, our report extends this discussion by specifically illustrating the possible role of Imipenem-Cilastatin-Relebactam plus Aztreonam in isolates that are phenotypically resistant to Ceftazidime-Avibactam plus Aztreonam, a scenario for which published clinical evidence remains particularly sparse.[12] At the same time, caution is warranted. Case reports cannot establish superiority, and success in individual patients may be influenced by inoculum burden, source control, host immune status,dosingstrategies,andthepresenceofadditional companion agents. Furthermore, evolving mechanisms such aspenicillin binding protein alterations, AmpCover expression, permeability defects, and target-site mutations may further erode the activity of Aztreonam-based regimens, emphasizing the importance of molecular characterization and repeat susceptibility testing during treatment.
Conclusion
From a therapeutic standpoint, our experience is aligned with the broader principles of the Infectious Diseases Society of America guidance, which supports Aztreonam-containing combinationsformetallo-β-lactamase-producing Enterobacteraleswhileacknowledgingthathigh-quality comparative clinical data remain limited. However, our report extends this discussion by specifically illustrating the possible role of Imipenem-Cilastatin-Relebactam plus Aztreonam in isolates that are phenotypically resistant to Ceftazidime-Avibactam plus Aztreonam, a scenario for which published 12clinical evidence remains particularly sparse.At the same time, caution is warranted. Case reports cannot establish superiority,andsuccessinindividualpatientsmaybe influenced by inoculum burden, source control, host immune status,dosingstrategies,andthepresenceofadditional companion agents. Furthermore, evolving mechanisms such aspenicillin-bindingprotein3alterations,AmpCover expression, permeability defects, and target-site mutations may further erode the activity of Aztreonam-based regimens, emphasizing the importance of molecular characterization and repeat susceptibility testing during treatment.
Consent and ethics
Written informed consent was obtained from the patient for publication.
References
- Zambrano-Calderero V, Vera-Álava F, Borges M. Risk factors associated with carbapenem-resistant Enterobacterales infections in hospitalized patients: a case-control study. Infez Med. 2026;34(1):96-106. doi:10.53854/liim-3401-10
- Sati H, Carrara E, Savoldi A, Hansen P, Garlasco J, et al; WHO Bacterial Priority Pathogens List Advisory Group. The WHO Bacterial Priority Pathogens List 2024: a prioritisation study to guide research, development, and public health strategies against antimicrobial resistance. Lancet Infect Dis. 2025;25(9):1033-1043. doi:10.1016/S1473-3099(25)00118-5
- Tompkins K, van Duin D. Treatment for carbapenem-resistant Enterobacterales infections: recent advances and future directions. Eur J Clin Microbiol Infect Dis. 2021;40(10):2053-2068. doi:10.1007/s10096-021-04296-1
- Fu Y, Zhu Y, Zhao F, Yao B, Yu Y, et al. In vitro synergistic and bactericidal effects of aztreonam in combination with ceftazidime/avibactam, meropenem/vaborbactam and imipenem/relebactam against dual-carbapenemase-producing Enterobacterales. Infect Drug Resist. 2024;17:3851-3861. doi:10.2147/IDR.S474150
- Tamma PD, Heil EL, Justo JA, Mathers AJ, Satlin MJ, Bonomo RA. Infectious Diseases Society of America 2024 guidance on the treatment of antimicrobial-resistant gram-negative infections. Clin Infect Dis. Published online August 7, 2024. doi:10.1093/cid/ciae403
- Boyd SE, Livermore DM, Hooper DC, Hope WW. Metallo-β-lactamases: structure, function, epidemiology, treatment options, and the development pipeline. Antimicrob Agents Chemother. 2020;64(10):e00397-20. doi:10.1128/AAC.00397-20
- Biagi M, Lee M, Wu T, Shajee A, Patel S, et al. Aztreonam in combination with imipenem-relebactam against clinical and isogenic strains of serine- and metallo-β-lactamase-producing Enterobacterales. Diagn Microbiol Infect Dis. 2022;103(2):115674. doi:10.1016/j.diagmicrobio.2022.115674
- Pipitò L, Rubino R, Immordino R, Bono E, Fasciana T, et al. Imipenem/relebactam plus aztreonam: first reported use in MDR Klebsiella pneumoniae sternal infection complicated by bacteremia. Antibiotics (Basel). 2025;14(10):1007. doi:10.3390/antibiotics14101007
- Simner PJ, Bergman Y, Conzemius R, Jacobs E, Tekle T, et al. An NDM-producing Escherichia coli clinical isolate exhibiting resistance to cefiderocol and the combination of ceftazidime-avibactam and aztreonam: another step toward pan-β-lactam resistance. Open Forum Infect Dis. 2023;10(7):ofad276. doi:10.1093/ofid/ofad276
- Senchyna F, Murugesan K, Rotunno W, Nadimpalli SS, Deresinski S, Banaei N. Sequential treatment failure with aztreonam-ceftazidime-avibactam followed by cefiderocol due to preexisting and acquired mechanisms in a New Delhi metallo-β-lactamase-producing Escherichia coli causing fatal bloodstream infection. Clin Infect Dis. 2024;78(6):1425-1428. doi:10.1093/cid/ciad759
- Karunanidhi A, Singh N, Watkins JV, Smith NM, Zang Y, et al. Ceftazidime/avibactam and aztreonam combined with an aminoglycoside combat metallo-β-lactamase-producing Klebsiella pneumoniae. Antimicrob Agents Chemother. 2026;70(3):e0154025. doi:10.1128/aac.01540-25
- Shields RK, Doi Y. Aztreonam combination therapy: an answer to metallo-β-lactamase-producing gram-negative bacteria? Clin Infect Dis. 2020;71(4):1099-1101. doi:10.1093/cid/ciz1159