Introduction
Hereditary angioedema (HAE) is a rare autosomal dominant disorder which is characterized by recurrent episodes of non-pruritic swelling involving the skin, gastrointestinal tract and upper airway. This is caused by dysregulation of the kallikrein–bradykinin pathway because of deficiency or dysfunction of C1-esterase inhibitor (C1-INH). [1] The estimated global prevalence of HAE ranges from approximately 1 in 50,000 to 1 in 100,000 individuals. [2] Clinically, HAE usually presents with episodic, asymmetric edema that commonly affects face, extremities or airway. One of the peculiar features of this edema is absence of urticaria or pruritus. Attacks of edema in cases of HAE may be precipitated by stress, trauma or infections and each attack usually last from several hours to days. Laryngeal involvement represents the most life-threatening manifestation and can result in fatal airway obstruction if not promptly recognized and treated. Because symptoms are nonspecific and frequently misinterpreted as allergic or idiopathic angioedema, diagnostic delays averaging several years have been reported in many cohorts.
The underlying pathophysiology of HAE involves quantitative or functional deficiency of C1-INH. [3] In the absence of adequate C1-INH activity, uncontrolled activation of plasma kallikrein leads to excessive production of bradykinin, a potent vasoactive peptide that increases vascular permeability and promotes localized edema formation. Unlike mast cell–mediated angioedema, bradykinin-mediated swelling does not respond to antihistamines, corticosteroids or epinephrine. This is crucial from point of appropriate management. Laboratory evaluation typically demonstrates reduced levels or functional activity of C1-INH along with decreased complement component C4. Genetic mutations in the SERPING1 gene account for the majority of these cases although a substantial proportion of patients may arise from de novo mutations without a clear family history. [4]
Factor V Leiden mutation represents one of the most common causes of inherited thrombophilia worldwide. It results from a single point mutation in the F5 gene which is responsible for production of a variant factor V molecule resistant to inactivation by activated protein C. This defect causes sustained thrombin thereby creating a hypercoagulable state that significantly increases the risk of venous thromboembolism in affected cases. The clinical features may include deep venous thrombosis (DVT) and pulmonary embolism (PE). While heterozygous carriers exhibit a moderate elevation in thrombotic risk individuals with homozygous mutations are at increased risk of serious thromboembolic events. Factor V Leiden prevalence varies geographically and affects approximately 3–8% of individuals of European descent but remaining relatively uncommon in other populations. Clinical management often requires long-term anticoagulation particularly in patients with history of recurrent thrombotic events. [5]
Both HAE and Factor V Leiden mutation involve dysregulation of plasma protease cascades within the complement, coagulation as well as kallikrein–kinin systems. These interconnected pathways play an important critical role in inflammation, vascular permeability and hemostasis. The coexistence of disorders affecting these systems in same individual may introduce additional clinical complexity affecting therapeutic decision-making. For example, treatment strategies for acute HAE attacks often include C1-INH replacement, bradykinin receptor antagonists or kallikrein inhibitors. However, management may become challenging in patients with concurrent prothrombotic conditions, particularly when therapies such as plasma infusion or invasive procedures are considered which may predispose individual to thrombotic events. Moreover, overlapping symptoms such as dyspnea, chest discomfort or edema may further complicate differentiation between angioedema attacks and thromboembolic complications.
Despite extensive literature describing hereditary angioedema and Factor V Leiden thrombophilia individually, reports describing their coexistence are exceedingly rare. The present report describes a 31-year-old patient diagnosed with hereditary angioedema due to C1-INH deficiency in the setting of homozygous Factor V Leiden mutation.
Case presentation
A 31-year-old woman presented with history of recurrent episodes of swelling which predominantly involved the right upper limb with extension to the face, eyelids and neck.
The swelling episodes were preceded by a flushing sensation accompanied by severe generalized body ache. She also reported episodes of abdominal pain without associated nausea, vomiting or alterations in bowel habits. During several attacks she experienced tachycardia and shortness of breath. The patient reported a history of similar recurrent episodes over the preceding years. These episodes generally involved the face, upper extremities, abdomen and genital region. There was a typical history of these episodes triggered by emotional stress. Attacks occurred approximately one to two times every week and resolved spontaneously within two to six hours without need of any specific treatment. Some episodes were reported to be associated with transient loss of consciousness.
Her past medical history was notable for recurrent venous thromboembolism. She had experienced multiple episodes of deep venous thrombosis and a pulmonary embolism which occurred four years earlier. Subsequent thrombophilia evaluation revealed a homozygous Factor V Leiden mutation and she had been managed accordingly. In addition, she had pulmonary arterial hypertension (PAH) for which she was receiving macitentan therapy. There was no known family history of similar swelling episodes or hereditary angioedema. On examination during an attack, the patient demonstrated non-pitting edema with mild erythema involving the right upper extremity. The swelling was non-pruritic and there was no associated lymphadenopathy. Respiratory examination revealed expiratory wheezing while cardiovascular and abdominal examinations were unremarkable (Figure 1).
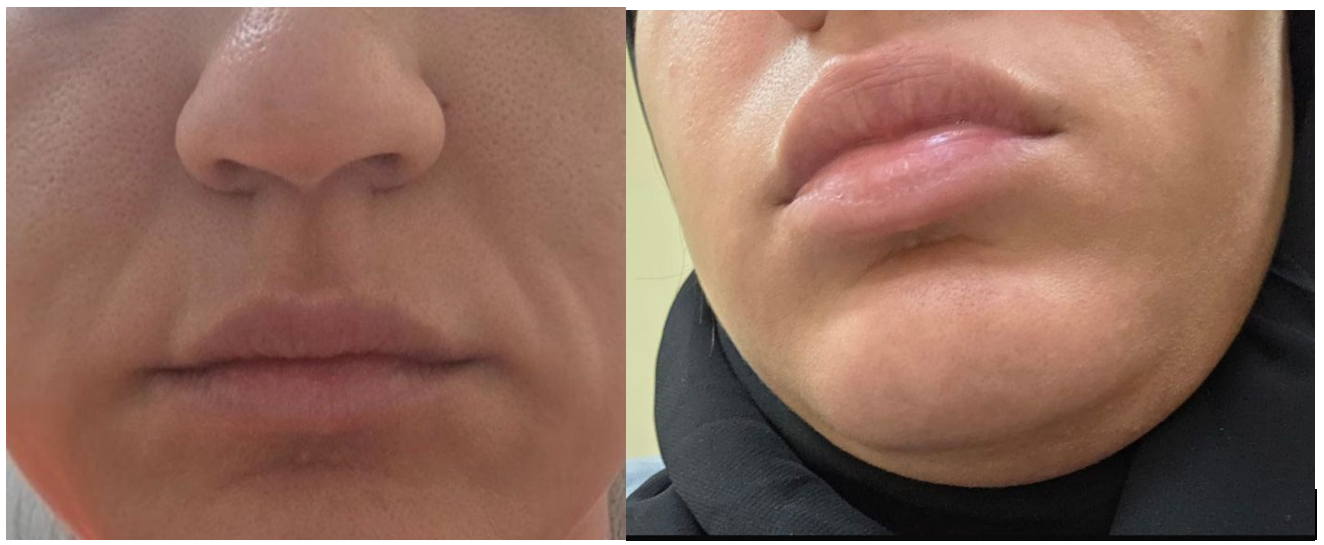
During observation, the swelling episode resolved spontaneously within approximately two hours. The edema did not respond to antihistamines including intramuscular chlorphenamine and both oral and intravenous diphenhydramine. Bronchodilator therapy provided no relief for the wheezing although transient improvement was noted following administration of epinephrine. The associated burning body pain was refractory to analgesics and improved only after administration of intravenous fentanyl and pethidine. Given the atypical presentation and recurrent episodes of swelling, an evaluation for mast-cell–mediated angioedema and systemic inflammatory conditions was undertaken. Laboratory investigations showed normal serum tryptase levels and negative antinuclear antibody (ANA) as well as antineutrophil cytoplasmic antibody (ANCA) tests. C-KIT mutation was also absent. These findings made mast-cell–mediated urticaria and systemic autoimmune etiologies unlikely.
Because of the recurrent episodes of non-pruritic swelling with airway involvement and poor response to antihistamines, hereditary angioedema (HAE) was suspected. Complement evaluation was therefore performed. Laboratory investigations demonstrated reduced C1-esterase inhibitor (C1-INH) levels (12 mg/dL) along with decreased complement component C4 levels, while C1q levels were within the normal range (233 mg/L). The combination of low C1-INH with reduced C4 and normal C1q supported the diagnosis of hereditary angioedema due to C1-INH deficiency rather than acquired angioedema. Fresh frozen plasma (FFP) transfusion was initially attempted as a therapeutic measure due to limited access to specific C1-INH replacement therapy. However, this resulted in worsening of pulmonary arterial hypertension and volume overload with no significant clinical improvement. Therefore, FFP transfusion was subsequently discontinued. Finally, a decision to do therapeutic plasmapheresis was made which resulted in significant symptomatic improvement and a reduction in the frequency of subsequent swelling episodes.
Discussion
Hereditary angioedema (HAE) due to C1-esterase inhibitor (C1-INH) deficiency is a rare but potentially life-threatening disorder. It is usually characterized by recurrent and self-limiting episodes of non-pruritic angioedema affecting the skin, gastrointestinal tract and upper airway. The clinical presentation of this patient—recurrent asymmetric swelling of the extremities and face and episodic abdominal pain along with absence of urticaria and lack of response to antihistamines—closely resembles the classical phenotype described in previous cohort studies on HAE. Bork et al reported that peripheral swelling and abdominal pain represent the most frequent manifestations of HAE. [6] The author emphasised diagnostic delays in these cases due to misinterpretation of clinical picture as allergic or idiopathic angioedema. Similarly, Longhurst et al emphasized that bradykinin-mediated angioedema typically fails to respond to conventional anti-allergic therapies. [7] This feature should prompt clinicians to evaluate complement levels and C1-INH function. In our patient the normal serum tryptase levels and negative autoimmune markers further supported a non–mast-cell-mediated mechanism. The eventual demonstration of low C1-INH levels confirmed the diagnosis of C1-INH deficiency–associated HAE. Clinicians should maintain a high index of suspicion for possibility of HAE in patients presenting with recurrent angioedema without urticaria.
Another important aspect highlighted by this case is the coexistence of HAE with inherited thrombophilia secondary to Factor V Leiden mutation. Although these conditions arise from different genetic abnormalities dysregulation of plasma protease systems is underlying cause in both. Various studies suggest that the complement and contact pathways closely interact with the coagulation cascade. Grover et al, in an observational study, demonstrated that patients with HAE have an increased risk of venous thromboembolism as compared to general population. [8] This finding suggest that chronic activation of the kallikrein–kinin system may influence coagulation pathways. Similarly, Sundler Björkman L et al identified an increased familial risk of venous thromboembolism among individuals with HAE supporting the concept that abnormalities in complement regulation may predispose to thrombotic complications. [9] In the present case, the patient had a documented history of recurrent deep venous thrombosis and pulmonary embolism due to homozygous Factor V Leiden mutation which represents a particularly high-risk thrombophilic state. The coexistence of these two conditions may therefore amplify both vascular permeability and thrombotic risk thereby adding complexity to clinical management.
Reports describing the simultaneous presence of hereditary angioedema and inherited thrombophilia remain extremely rare. One such notable case was described by Celikel et al In this patient hereditary angioedema as well as heterozygous Factor V Leiden mutation was present. Patient developed purpura fulminans after hormonal therapy. [10] The authors suggested that the coexistence of complement dysregulation and thrombophilia may have predisposed patient to severe vascular complications including purpura fulminans. Although our patient did not develop serious complication such as purpura fulminans there is always a potential for complex clinical interactions between these disorders.
Management of HAE in patients with coexisting comorbidities may present significant therapeutic challenges. Current international consensus guidelines recommend C1-INH replacement therapy, bradykinin B2-receptor antagonists such as Icatibant or plasma kallikrein inhibitors for management of acute attacks. However, in many clinical settings these agents are not readily available thereby necessitating use of alternative, non-specific therapies such as administration of fresh frozen plasma (FFP). While FFP provides exogenous C1-INH it also contains substrates that may potentially exacerbate bradykinin generation. Cicardi and Zanichelli have noted that although FFP may be beneficial in some cases of HAE, its use can occasionally worsen symptoms secondary to activation of complement or contact pathways. In our patient, administration of FFP was associated with worsening pulmonary arterial hypertension and volume overload without symptomatic improvement requiring discontinuation. Subsequently therapeutic plasmapheresis was done in this patient that resulted in marked clinical improvement and reduction in subsequent attacks.
Conclusion
Presence of HAE due to C1-INH deficiency and thrombophilia secondary to homozygous Factor V Leiden mutation in same individual represent a rare clinical possibility. These disorders are caused by dysregulation of inflammatory and coagulation pathways. This case underscores the importance of considering the possibility of bradykinin-mediated angioedema in patients with recurrent edema particularly if it is non-pruritic and unresponsive to antihistamines. Awareness of such coupled with a high index of suspicion will help in facilitating early recognition and guiding individualized therapeutic strategies in patients of HAE with concurrent prothrombotic disorders such as thrombophilia caused by homozygous Factor V Leiden mutation.
Consent and ethics
Written informed consent was obtained from the patient for publication.
References
- Maurer M, Magerl M, Betschel S, Aberer W, Ansotegui IJ, Aygören-Pürsün E, et al. The international WAO/EAACI guideline for the management of hereditary angioedema—the 2021 revision and update. Allergy. 2022;77(7):1961-1990.
- Ghazi A, Grant JA. Hereditary angioedema: epidemiology, management, and role of icatibant. Biologics. 2013;7:103-13. doi: 10.2147/BTT.S27566. Epub 2013 May 3.
- Kaplan AP, Joseph K. Pathogenic mechanisms of bradykinin mediated diseases: dysregulation of an innate inflammatory pathway. Adv Immunol. 2014;121:41-89. doi: 10.1016/B978-0-12-800100-4.00002-7.
- Ponard D, Gaboriaud C, Charignon D, Ghannam A, Wagenaar-Bos IGA et al. SERPING1 mutation update: Mutation spectrum and C1 Inhibitor phenotypes. Hum Mutat. 2020 Jan;41(1):38-57. doi: 10.1002/humu.23917.
- Kujovich JL. Factor V Leiden thrombophilia. Genet Med. 2011 Jan;13(1):1-16. doi: 10.1097/GIM.0b013e3181faa0f2.
- Bork K, Meng G, Staubach P, Hardt J. Hereditary angioedema: new findings concerning symptoms, affected organs, and course. Am J Med. 2006 Mar;119(3):267-74. doi: 10.1016/j.amjmed.2005.09.064.
- Longhurst H, Bork K. Hereditary angioedema: causes, manifestations and treatment. Br J Hosp Med (Lond). 2019;80(7):391-398.
- Grover SP, Sundler Björkman L, Egesten A, Moll S, Mackman N. Hereditary angioedema is associated with an increased risk of venous thromboembolism. J Thromb Haemost. 2022 Nov;20(11):2703-2706. doi: 10.1111/jth.15870. Epub 2022 Sep 14.
- Sundler Björkman L, Pirouzifard M, Grover SP, Egesten A, Sundquist J, Sundquist K, Zöller B. Increased risk of venous thromboembolism in young and middle-aged individuals with hereditary angioedema: a family study. Blood. 2024 Jul 25;144(4):435-444. doi: 10.1182/blood.2023022996.
- Celikel S, Buyukasik Y, Karakaya G, Kalyoncu AF. Hereditary angioedema associated with heterozygous factor V Leiden mutation in a patient with Purpura fulminans. Int Arch Allergy Immunol. 2007;142(2):175-8. doi: 10.1159/000096611. Epub 2006 Oct 27. PMID: 17068406.