Introduction
Endometriosis is a chronic, estrogen-dependent inflammatory disorder defined by the presence of endometrial glands and stroma outside the uterine cavity. Although typically pelvic in distribution, ectopic implants may occur at extrapelvic locations, including the abdominal wall, where they can form discrete endometriotic nodules within subcutaneous tissue, fascia, and muscle. Abdominal wall endometriosis (AWE) is uncommon yet clinically important and is frequently associated with prior obstetric or gynecologic surgery. It is most commonly seen after a cesarean section which has risen globally over recent decades. In this demographic, the burden of symptoms can be substantial: patients commonly experience cyclical pain that interferes with daily activity and quality of life, and delays in diagnosis may prolong morbidity and increase healthcare utilization. AWE, while uncommon, should be considered in any reproductive-aged woman presenting with a painful abdominal wall mass in proximity to a surgical scar. AWE is more likely if symptoms are reported to be fluctuating with menses.[1]
Scar endometriosis represents the most recognized subset of AWE and is thought to arise largely from iatrogenic implantation of endometrial tissue at the time of uterine surgery. During cesarean delivery or other surgical procedures endometrial cells can be implanted at the wound edges. These cells subsequently proliferate under cyclical ovarian hormonal stimulation. Other than direct implantation proposed contributing mechanisms include altered local immune surveillance and metaplastic change within susceptible tissues. This may help explain cases occurring at sites remote from a uterine incision or after non-gynecologic operations. It is interesting to note that the clinical latency between surgery and symptom onset is variable, and the disease can present months to years after the inciting procedure. This latent period makes it difficult to establish a causal relationship between uterine surgery and endometriosis.[2]
Clinically, scar endometriosis is classically associated with the triad of (i) a palpable mass, (ii) cyclic pain, and (iii) a history of prior surgery; however, real-world presentations are often incomplete or atypical. Pain may be focal, deep, or neuropathic in character; swelling may be subtle; and the lesion may be mistaken for incisional hernia, suture granuloma, abscess, lipoma, desmoid tumor, or other soft-tissue neoplasm. In some series, a sizeable proportion of patients do not demonstrate the full “classic” symptom triad, and the absence of visible skin changes can further mislead clinicians. Consequently, diagnostic delay is common, with repeated empiric treatments or referrals across specialties before definitive evaluation.[3]
Ultrasonography is typically the first-line modality because of its ability to evaluate the relationship of a lesion to the fascia and to exclude common differentials such as hernia and other subcutaneous lesions. Computed tomography or magnetic resonance imaging (MRI) can better delineate lesion extent, depth of infiltration and involvement of the rectus sheath. MRI is particularly useful when lesions demonstrate hemorrhagic components, and it may improve characterization of complex masses; nonetheless, imaging findings are not pathognomonic, and overlap exists with benign and malignant soft-tissue processes. Ultimately, histopathologic confirmation remains definitive and show endometrial glands and stroma (often with hemorrhage and hemosiderin-laden macrophages) within fibrous or muscular tissue. Authoritative clinical reviews emphasize that while imaging guides suspicion and surgical strategy, tissue diagnosis is required to confirm AWE and to exclude rare malignant transformation in long-standing disease.[4]
Management is centred on complete surgical excision with clear margins, which is widely regarded as the most effective approach for symptom resolution and recurrence prevention. Medical therapies (e.g., progestins, danazol, or gonadotropin-releasing hormone agonists) may provide temporary symptom control in select patients but tend to yield incomplete responses and frequent recurrence once discontinued; moreover, they do not provide histologic confirmation or definitively rule out malignancy. Surgical series and clinical updates consistently report high rates of symptomatic improvement after wide local excision, with recurrence largely linked to incomplete resection or unrecognized multifocal disease, and larger/deeper lesions may require mesh repair or complex abdominal wall reconstruction. Despite this, clinically meaningful knowledge gaps persist: scar endometriosis remains underdiagnosed, standardized diagnostic pathways are not uniformly applied across surgical and gynecologic settings, and the literature is dominated by small series and case reports with heterogeneous imaging protocols, margin definitions, and follow-up intervals. By presenting a well-characterized case with cyclical scar-site pain, supportive MRI findings, complete excision, and histopathologic confirmation, the present report aims to reinforce practical diagnostic cues, highlight operative principles that reduce recurrence, and contribute incremental evidence toward improved recognition and management of this rare but impactful surgical sequela.[5]
Case presentation
A 33-year-old multiparous woman (G4P3+1; living 4) presented with cyclical periumbilical abdominal pain that had progressively worsened over time and was consistently exacerbated during menstruation. Her past medical history showed that she was diagnosed to be having epilepsy at four years of age. She was well controlled for the preceding two years on levetiracetam (Keppra) 1 g at bedtime (HS) and carbamazepine (Tegretol) 400 mg HS. She also had bronchial asthma managed with salbutamol inhaler (Ventolin) as needed (PRN). In addition, she had hypothyroidism as well as prolactinoma; she had previously received levothyroxine for seven years but had more recently been taking selenium and reported clinical improvement over the past year.
Her obstetric history included three prior cesarean sections. The most recent cesarean section was done 2 years back resulting in twin births. She also had a left ectopic pregnancy treated surgically with left salpingectomy in approximately 4 years back. Her additional surgical history included an inguinal hernia repair in 2023 and an open umbilical hernia repair in 3 years back.
On examination, a well-defined, tender nodule was palpated in the left lower abdominal wall, approximately 5 cm lateral to the midline and 5 cm above the cesarean scar. No other abnormalities were detected on systemic examination. Pelvic magnetic resonance imaging (MRI) demonstrated presence of enhancing lesion that measured 6.2 × 3.4 × 2.2 cm within the left lower abdominal wall. It was characterized by iso–T2 and high T1 signal intensity with diffusion restriction. These MRI findings were suggestive of abdominal wall endometriosis. Additionally, partially imaged soft-tissue thickening in the umbilical region raised suspicion of extrapelvic endometriosis. Laboratory investigations were within normal limits. Based on the characteristic cyclical nature of symptoms, prior surgical history and MRI features a provisional diagnosis of scar endometriosis was made and the patient was planned for surgical excision.
Intraoperatively, a firm, well-circumscribed mass measuring 5 × 3 × 2 cm was found immediately beneath the rectus sheath. It was located 5 cm lateral to the midline and 4–5 cm above the cesarean section scar, just inferior to the umbilicus. On inspection, the lesion contained dark, “chocolate-colored” material. The mass was excised in its entirety while preserving the rectus muscle and peritoneum (Figure 1).
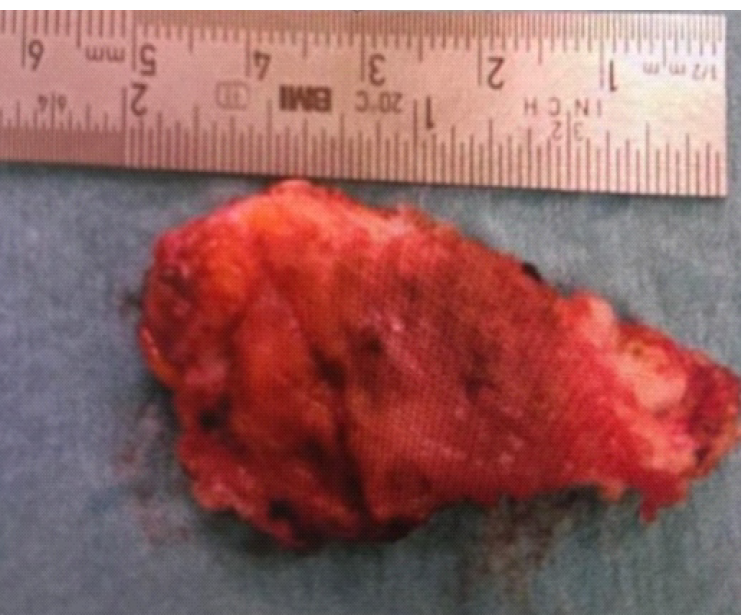
Histopathological examination was done which showed presence of characteristic fibrofatty tissue admixed with skeletal muscle fibers. Additionally, foci of endometrial glands with surrounding stroma were also seen. These histopathological features confirmed the diagnosis of scar endometriosis.
The postoperative course of the patient was uneventful. She was discharged on postoperative day 2 and remains under regular follow-up. Till 6 months postoperatively she did not report any signs or symptoms that may suggest recurrence.
Discussion
Abdominal wall endometriosis (AWE) remains an underrecognized manifestation of extrapelvic endometriosis, yet it carries a characteristic clinicopathologic link with prior uterine surgery that is highly relevant to the present case. Our patient—33 years old with three prior cesarean deliveries and additional abdominal operations—fits the demographic and exposure profile described in larger syntheses of AWE. In the systematic review by Horton et al, most patients were in their early thirties and commonly presented years after an index uterine surgery. [6] The study underscored both the reproductive-age predilection and the clinically important latency between implantation and symptomatic disease. In parallel, the large cohort and literature review by Zhang et al on cesarean scar endometriosis (CSE) reinforces the iatrogenic implantation hypothesis by demonstrating that the majority of cases arise after cesarean delivery and that latency and symptom duration can be measured in months to years, rather than weeks. [7] Our patient's cyclic pain beginning after her last cesarean section is therefore consistent with the prevailing model in which ectopic endometrial glands and stroma—introduced into the abdominal wall during uterine surgery—proliferate under hormonal influence and generate localized inflammation, hemorrhage, and fibrosis. Importantly, our case also raises the possibility of multifocal extrapelvic disease (umbilical soft-tissue thickening on MRI), aligning with contemporary observations that AWE can occasionally coexist with lesions beyond the primary scar region and may not be strictly confined to the surgical incision line.
Although the “classic triad” oefn dometriosis consisst of cyclical pain, palpable mass and prior surgery, this may not be present in all the cases. Many cases often deviate from this template contributing to repeated misdiagnosis and delayed management. Our patient had a well-defined tender nodule and striking catamenial exacerbation, which strengthened clinical suspicion; however, its location (lateral and above the Pfannenstiel scar) and her history of non-gynaecologic abdominal wall repairs could reasonably broaden the differential toward hernia recurrence, suture granuloma, desmoid-type fibromatosis, or other soft-tissue pathology. In the 10-year experience reported by Bektaş et al, patients commonly underwent evaluation for alternative surgical diagnoses before endometriosis was confirmed. [8] This reflects how referral pathways often begin in general surgery rather than gynecology. Similarly, the multicenter observational series by Benedetto et al (83 cases) highlighted that AWE can occur after both obstetric as well as laparoscopic procedures. [9] In our case, the combination of catamenial pain and a discrete abdominal wall nodule was diagnostically valuable and supports the clinical message that symptom periodicity should be weighted heavily. This is important when the lesion's topography is not directly within the scar line and may mislead the treating physician into other diagnoses. The operative finding of “chocolate-colored” material further corroborates the recurrent hemorrhage and hemosiderin deposition that typically accompany long-standing endometriotic implants.
Imaging is pivotal for defining lesion extent and surgical planning, but it must be interpreted within the clinical context because no single radiologic feature is pathognomonic. Our patient's MRI demonstrated a lesion with high T1 signal and diffusion restriction—features compatible with hemorrhagic content and high cellularity/fibrosis—leading to a strong preoperative impression of AWE. A study by Hensen et al emphasized that abdominal wall endometriosis appears as solid hypoechoic vascular mass on ultrasound. [10] Further the authors stated that CT/MRI can be used selectively to clarify deep extension or complex anatomy. Extending this, Busard et al specifically characterized MRI appearances of AWE and described how lesions may appear heterogeneous with foci of high signal intensity related to hemorrhage on T1-weighted sequences, and variable T2 signal due to fibrosis and repeated bleeding. [11] Our imaging findings—T1 hyperintensity with iso–T2 signal—fit well within these reported patterns and were clinically useful in anticipating a lesion near the rectus sheath. A notable point in our report is the discrepancy between the MRI-reported measurement and the intraoperative specimen size, which can occur when surrounding edema, fibrotic reaction, or partially imaged contiguous foci inflate radiologic dimensions compared with the dominant resected nodule.
While hormonal therapy including gonadotropin-releasing hormone agonists, danazol and progestins has been explored as a treatment option in many cases its effectiveness in AWE is limited. Medical therapy may provide temporary symptom relief and slight lesion regression but recurrence is common upon discontinuation.
Wide local excision remains the crucial aspect of management. If done meticulously it will cause symptom resolution as well as reduce the chances of future recurrence. In our case, complete excision was achieved with preservation of rectus muscle and peritoneum and the patient remained symptom-free at 6 months. While this short-term course is reassuring longer follow-up is important because recurrence can occur after an initially successful resection. The 11-year cohort study by Marras et al reported that there is a significant diagnostic delay in cases of endometriosis. [12] The authors also documented recurrence over extended follow-up periods. These findings emphasize that clinical cure should be assessed over years rather than months. Moreover, Wu et al studied a large surgical cohort and proposed a depth-based classification (superficial/subcutaneous, fascial-muscular, and peritoneal involvement). [13]
Although malignant transformation is uncommon. There is need for histologic confirmation and vigilance for atypical features in cases of endometriosis. These atypical features may include rapid growth, non-cyclic pain, ulceration, fixation or presence of lymphadenopathy. In the large recurrence-focused retrospective study by Chen et al postoperative relapse occurred in a measurable minority of patients. [14] Recurrence was associated with factors such as multiple cesarean deliveries and operative complexity. Mert et al described clear cell carcinoma arising in abdominal wall scars in association with endometriotic implants. [15] These cases shows that, though very rare, malignant transformation can occur in cases of endometriosis and it can be aggressive and may require multimodal oncologic management.
Conclusion
Abdominal wall endometriosis is an uncommon but important cause of abdominal wall pain and nodules after cesarean delivery, and it should remain high on the differential when pain is clearly catamenial. This case highlights that lesions may occur above and lateral to the scar line, creating diagnostic confusion with hernia, granuloma or soft-tissue tumors. Imaging such as ultrasound and MRI can support diagnosis however the definitive diagnosis can only be made on the basis of histopathology. Wide local excision with histopathologic confirmation remains definitive. Longer follow-up is required to detect recurrence and rare malignant change.
Consent and ethics
Written informed consent was obtained from the patient for publication.
References
- Foley CE, Ayers PG, Lee TT. Abdominal Wall Endometriosis. Obstet Gynecol Clin North Am. 2022 Jun;49(2):369-380. doi: 10.1016/j.ogc.2022.02.013.
- Carsote M, Terzea DC, Valea A, Gheorghisan-Galateanu AA. Abdominal wall endometriosis (a narrative review). Int J Med Sci. 2020 Feb 10;17(4):536-542. doi: 10.7150/ijms.38679.
- Grigore M, Socolov D, Pavaleanu I, Scripcariu I, Grigore AM, Micu R. Abdominal wall endometriosis: an update in clinical, imagistic features, and management options. Med Ultrason. 2017 Nov 29;19(4):430-437. doi:10.11152/mu-1248. PMID: 29197920.
- Bektaş H, Bilsel Y, Sari YS, Ersöz F, Koç O, Deniz M, Boran B, Huq GE. Abdominal wall endometrioma; a 10-year experience and brief review of the literature. J Surg Res. 2010 Nov;164(1):e77-81. doi:10.1016/j.jss.2010.07.043. Epub 2010 Aug 15.
- Triantafyllidou O, Mili N, Kalampokas T, Vlahos N, Kalampokas E. Surgical management of abdominal wall sheath and rectus abdominis muscle endometriosis: a case report and literature review. Front Surg. 2024 Jan 11;10:1335931. doi:10.3389/fsurg.2023.1335931.
- Horton JD, DeZee KJ, Ahnfeldt EP, Wagner M. Abdominal wall endometriosis: a surgeon's perspective and review of 445 cases. Am J Surg. 2008;196(2):207-212. doi:10.1016/j.amjsurg.2007.07.035.
- Zhang P, Sun Y, Zhang C, Yang Y, Zhang L, Wang N, Xu H. Cesarean scar endometriosis: presentation of 198 cases and literature review. BMC Womens Health. 2019;19(1):14. doi:10.1186/s12905-019-0711-8.
- Bektaş H, Bilsel Y, Sari YS, et al. Abdominal wall endometrioma; a 10-year experience and brief review of the literature. J Surg Res. 2010;164(1):e77-e81. doi:10.1016/j.jss.2010.07.043.
- Benedetto C, Cacozza D, de Sousa Costa D, et al. Abdominal wall endometriosis: report of 83 cases. Int J Gynaecol Obstet. 2022;159(2):530-536. doi:10.1002/ijgo.14167.
- Hensen JH, Van Breda Vriesman AC, Puylaert JB. Abdominal wall endometriosis: clinical presentation and imaging features with emphasis on sonography. AJR Am J Roentgenol. 2006 Mar;186(3):616-20. doi:10.2214/AJR.04.1619. PMID: 16498086.
- Busard MPH, Mijatovic V, van Kuijk C, Hompes PGA, van Waesberghe JHTM. Appearance of abdominal wall endometriosis on MR imaging. Eur Radiol. 2010;20(5):1267-1276. doi:10.1007/s00330-009-1658-1.
- Marras S, Pluchino N, Petignat P, Wenger JM, Ris F, Buchs NC, Dubuisson J. Abdominal wall endometriosis: an 11-year retrospective observational cohort study. Eur J Obstet Gynecol Reprod Biol X. 2019;4:100096. doi:10.1016/j.eurox.2019.100096.
- Wu Y, Dai Y, Zhang J, et al. The clinical features and long-term surgical outcomes of different types of abdominal wall endometriosis. Arch Gynecol Obstet. 2023;307(1):163-168. doi:10.1007/s00404-022-06579-0.
- Chen Y, Dong Y, Hu H, Zhou Y, Xue M, Li D, Sun L. Analysis of related factors for postoperative recurrence of abdominal wall endometriosis: a retrospective cohort study. BMC Womens Health. 2025;25(1):548. doi:10.1186/s12905-025-03984-w.
- Mert I, Semaan A, Kim S, et al. Clear cell carcinoma arising in the abdominal wall: two case reports and literature review. Am J Obstet Gynecol. 2012;207(2):e7-e9. doi:10.1016/j.ajog.2012.05.029.